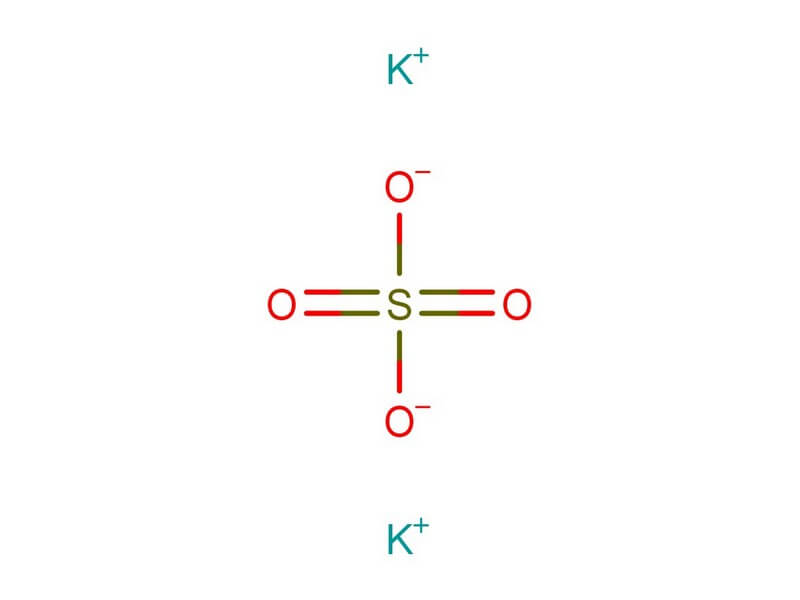
Synthesis reaction(include an example):A chemical reaction in which two or more substances react to form a single substance. What is the purpose of an empirical formula? 2. Chapter 7: Chemical Nomenclature 7.1 Ionic Compounds Lesson Review Questions Questions 1.Atoms have an oxidation number of zero in a (n) Jun 15, 2016 distribution of electrons among the bonded particles in a compound. type of bond holding particles together in a compound. Read Book Chapter 7 Section 2 Chemical Formulas And Compounds Chapter 7 Section 2 Chemical Formulas And Compounds.If you want to witty books, lots of novels, tale, jokes, and more12 hours ago Nonetheless, this .Chemical Formulas And Compounds Chapter 7 Review Answer Key If you ally obsession such a referred Chemical Formulas And Compounds Chapter 7 Review Answer Key book that will offer you worth, get the no question best seller from us currently from several preferred authors. There is an explosive reaction which produces water. Mix one part oxygen with two parts hydrogen, stand well back and ignite. Cross over the charges by using the absolute value of each ion’s charge as the subscript for the other ion: Al 2 3+ O 3 2− 3.02 Jul,2015. Write the symbols for the ions side by side. 6.Ionic Binary Compounds 7-1-2 Determine the formula of an ionic compound formed between two given ions. Subscript: A number written lower in a chemical formula indicating the amount of atoms present of an element (this number is not to be changed once written in chemical formula). 
Dewey 1994Coefficient: Numbers that appear before a formula in a chemical equation to show the relative proportions of each reactant and product. This is why you remain in the best website to look the unbelievable book to have. 8) beryllium bicarbonate.As this Chemical Formulas And Compounds Chapter 7 Review Answers, it ends taking place innate one of the favored ebook Chemical Formulas And Compounds Chapter 7 Review Answers collections that we have. Write the formulas for the following chemical compounds. Chemical formula Worksheet Answers Awesome Chemical Reaction Gizmo from chemical names and formulas worksheet answers, …Naming Compounds Test Review Practice.Cross over the charges by using the absolute value of each ion’s charge as the subscript for the other ion: Al 2 3+ O 3 2− 3.Apr 6, 2021 Section 7.1 – Chemical Names and Formulas HW – Notes on section 7.1 pgs Objectives Students will be able to : Explain the significance of a chemical formula .Ionic Binary Compounds 7-1-2 Determine the formula of an ionic compound formed between two given ions. 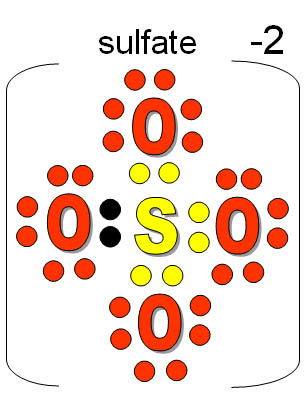
In Chapter 7, students will learn how to write the chemical formula of any given compound, either ionic or molecular, given that compound's. It serves as a summary of all course content discussed so far, as the focus of this chapter is on process skills rather than new concepts or theory.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
